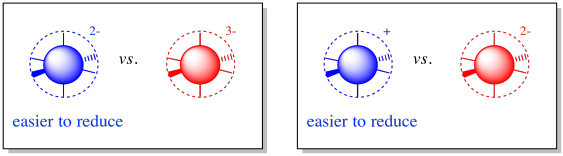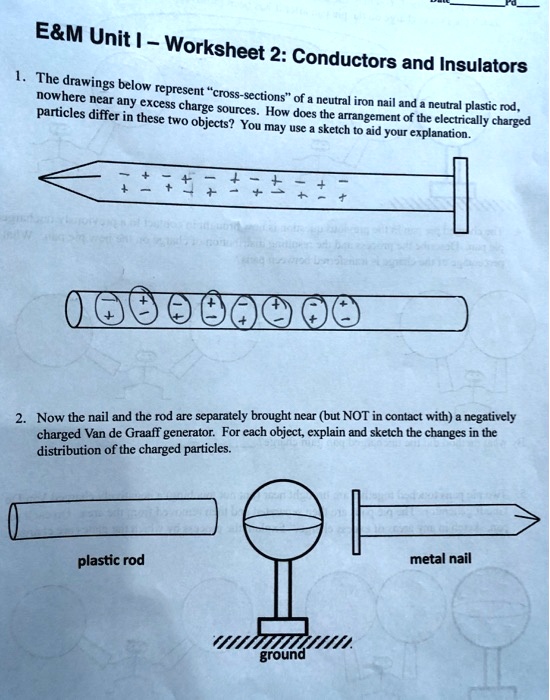
SOLVED: E M Unit Worksheet 2: Conductors and Insulators The drawings below represent nowhere near any excess = cross-sections' charge = neutral iron nail and particles differ sources How does the neutral

Question Video: Deducing the Ionic Formula of an Ionic Compound Where Both Ions Have Greater-Than-One Charge | Nagwa
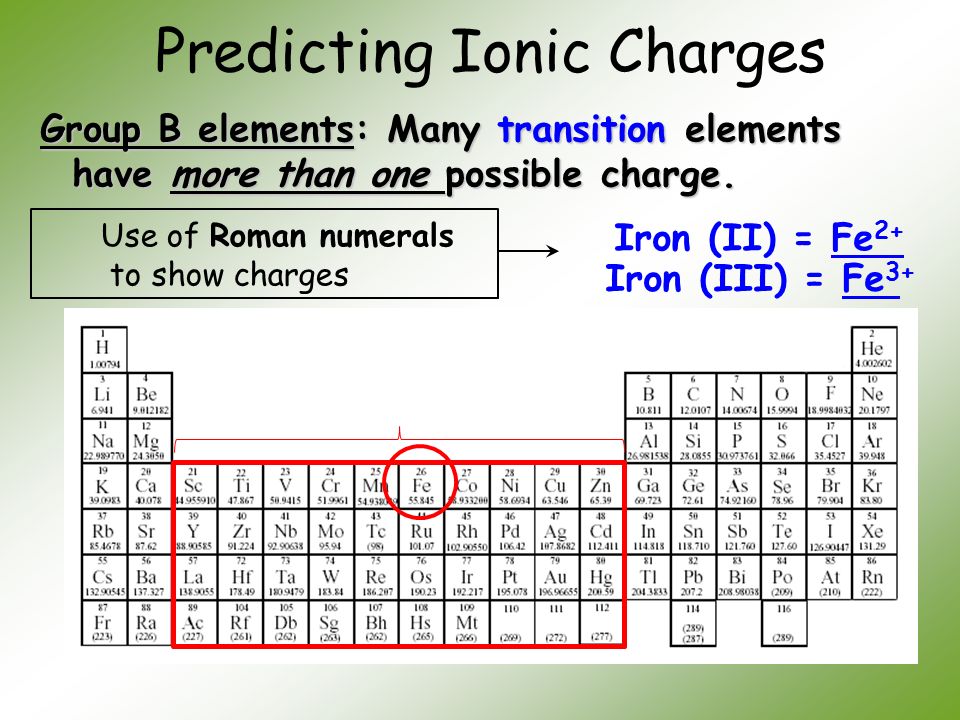
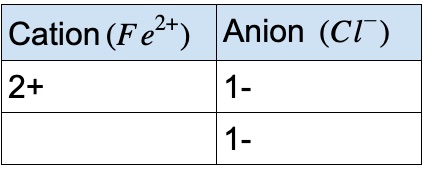
Warm Up Iron can form two different oxides ….. Fe 2 O 3 and FeO. We know for absolute certainty that an oxygen anion always has a -2 charge. What is going. - ppt download

Draw the step by step structures to obtain the Lewis dot structures and balance the charges of the following. a.) Fe_2S_3 b.) Mg_3N_2 | Homework.Study.com
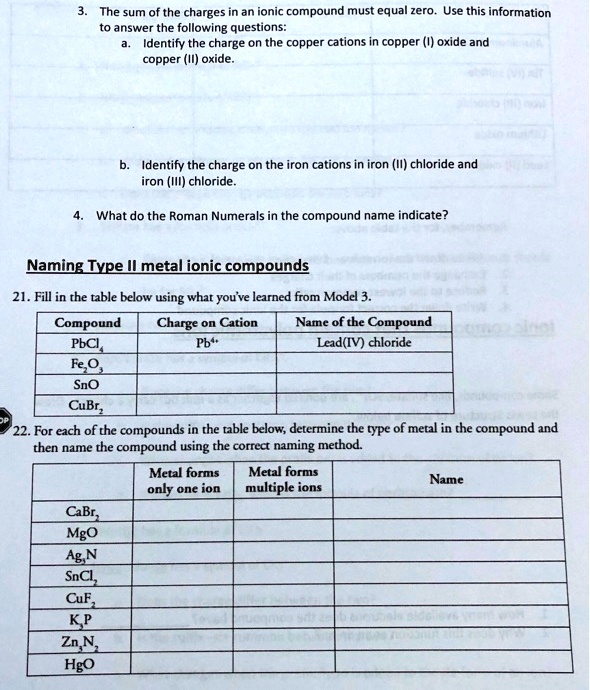
SOLVED: The sum of the charges in an ionic compound must equal zero. Use this information to answer the following questions: Identify the charge on the copper cations in copper (I) oxide
![PDF] Effect of Iron Oxides on Positive and Negative Charges in Clays and Soils | Semantic Scholar PDF] Effect of Iron Oxides on Positive and Negative Charges in Clays and Soils | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0119bf4c9df51dcf59f46ab945694cb409e711f8/5-Table2-1.png)






:max_bytes(150000):strip_icc()/PeriodicTableCharge-WBG-56a12db23df78cf772682c37.png)