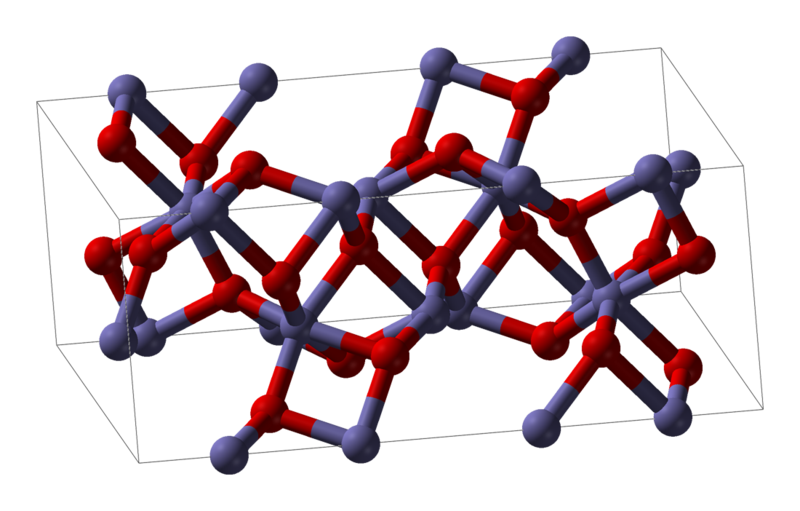
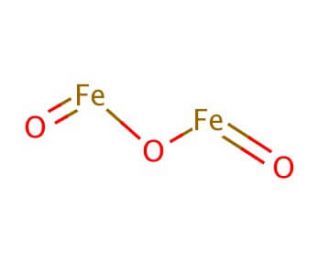
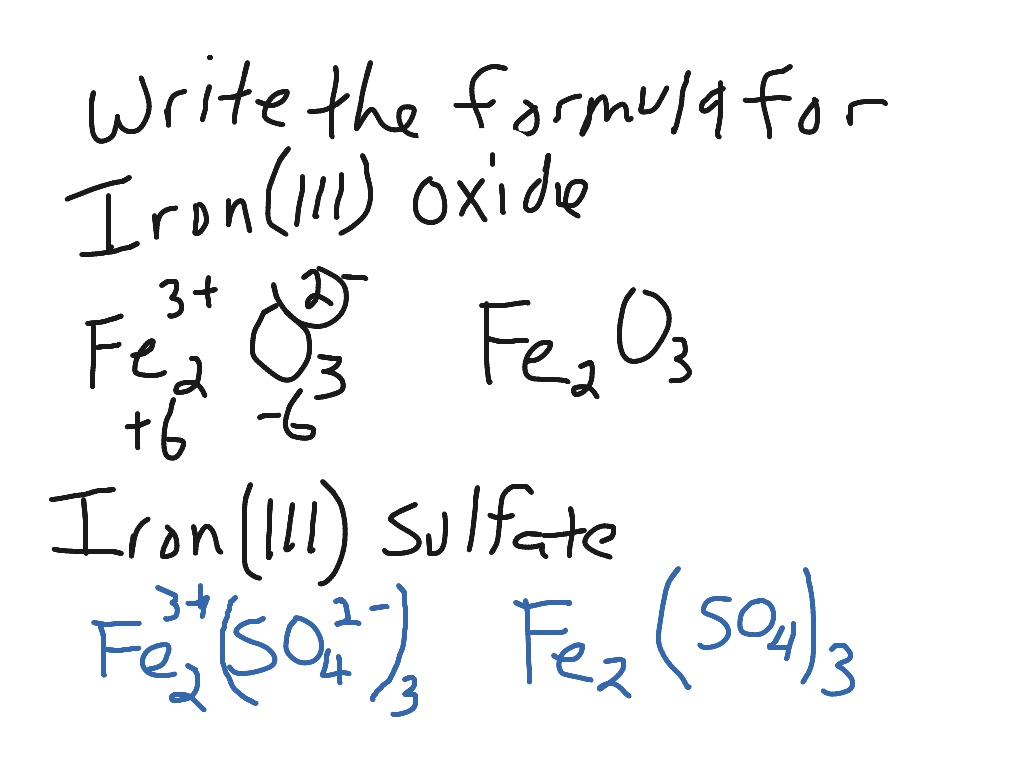_how-to-write-the-formula-for-iron-iii-oxide.jpg)
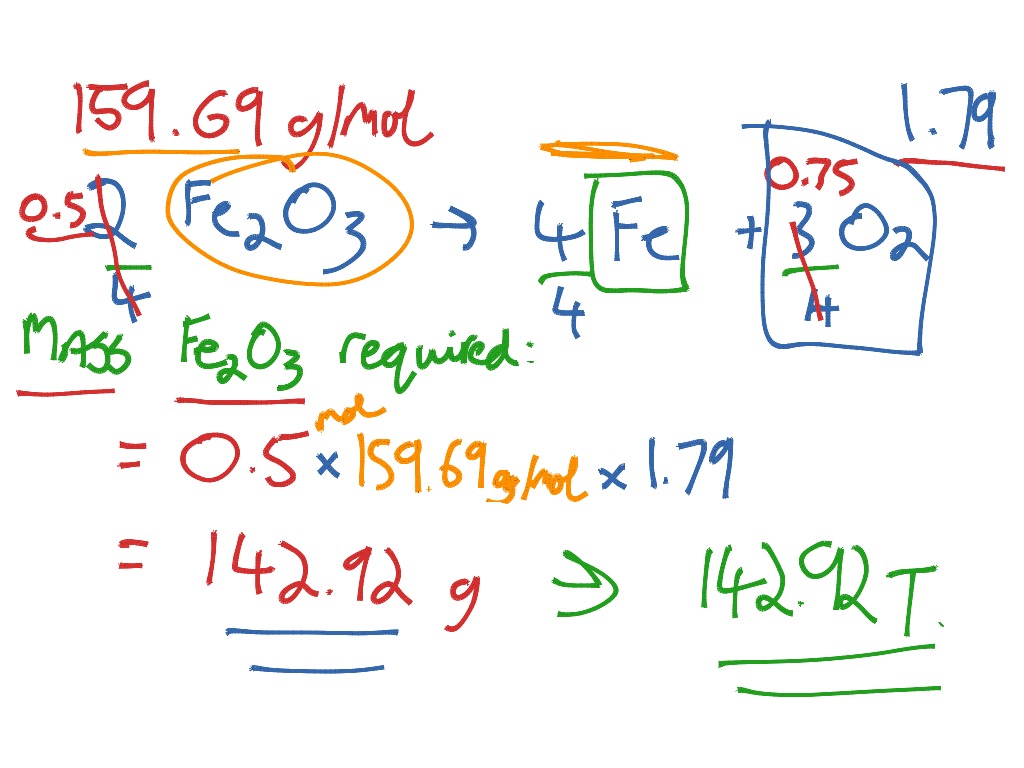
Determine the empirical formula of an oxide of iron, which has 69.9% iron and 30.1% dioxygen by mass from ferric oxide formula Watch Video - HiFiMov.co

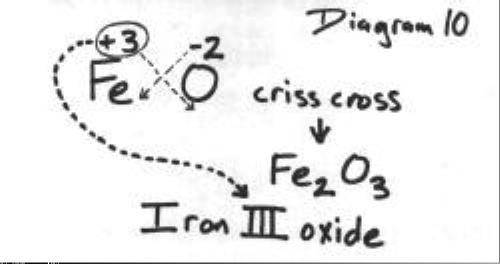
Question Video: Deducing the Ionic Formula of an Ionic Compound Where Both Ions Have Greater-Than-One Charge | Nagwa