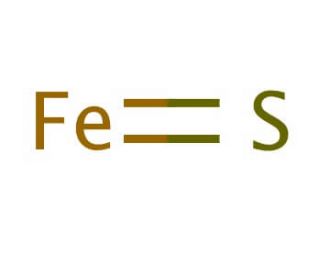Molecules and Compounds: Nomenclature. 2 CompoundsCompounds vs. Elements Compound 1: Table Salt Properties: Soluble crystals, stable, edible Elements. - ppt download
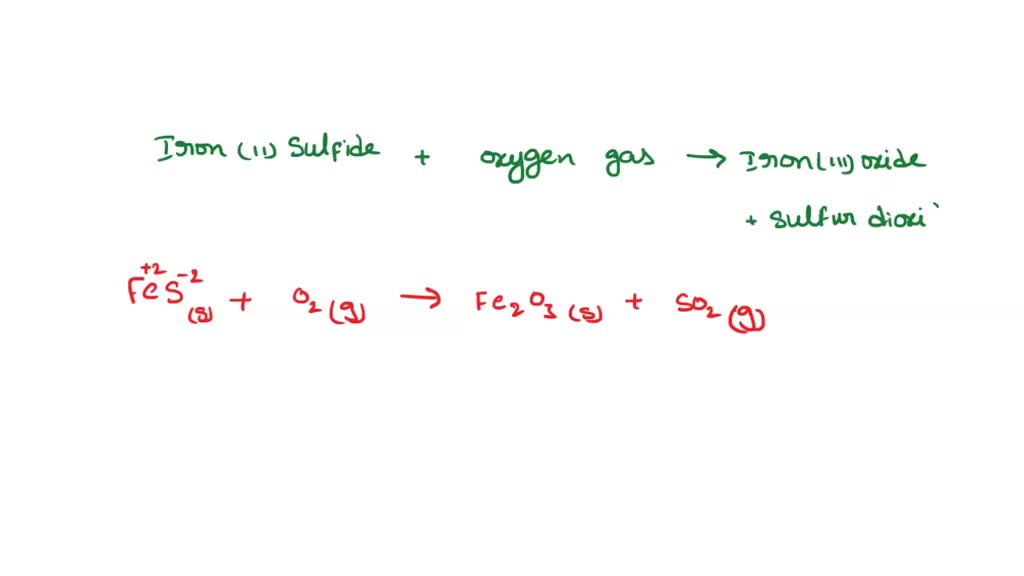
SOLVED: A student reacted iron (II) sulfide with oxygen gas to form iron (III) oxide and sulfur dioxide. What is the balance equation? 4FeS(s) + 7020) 2Fe203(s) 4502() AFeS(s) + 402lg) 2Fe203(s)
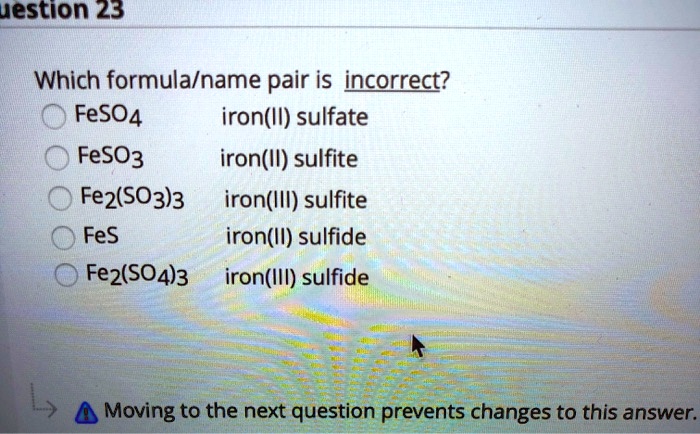
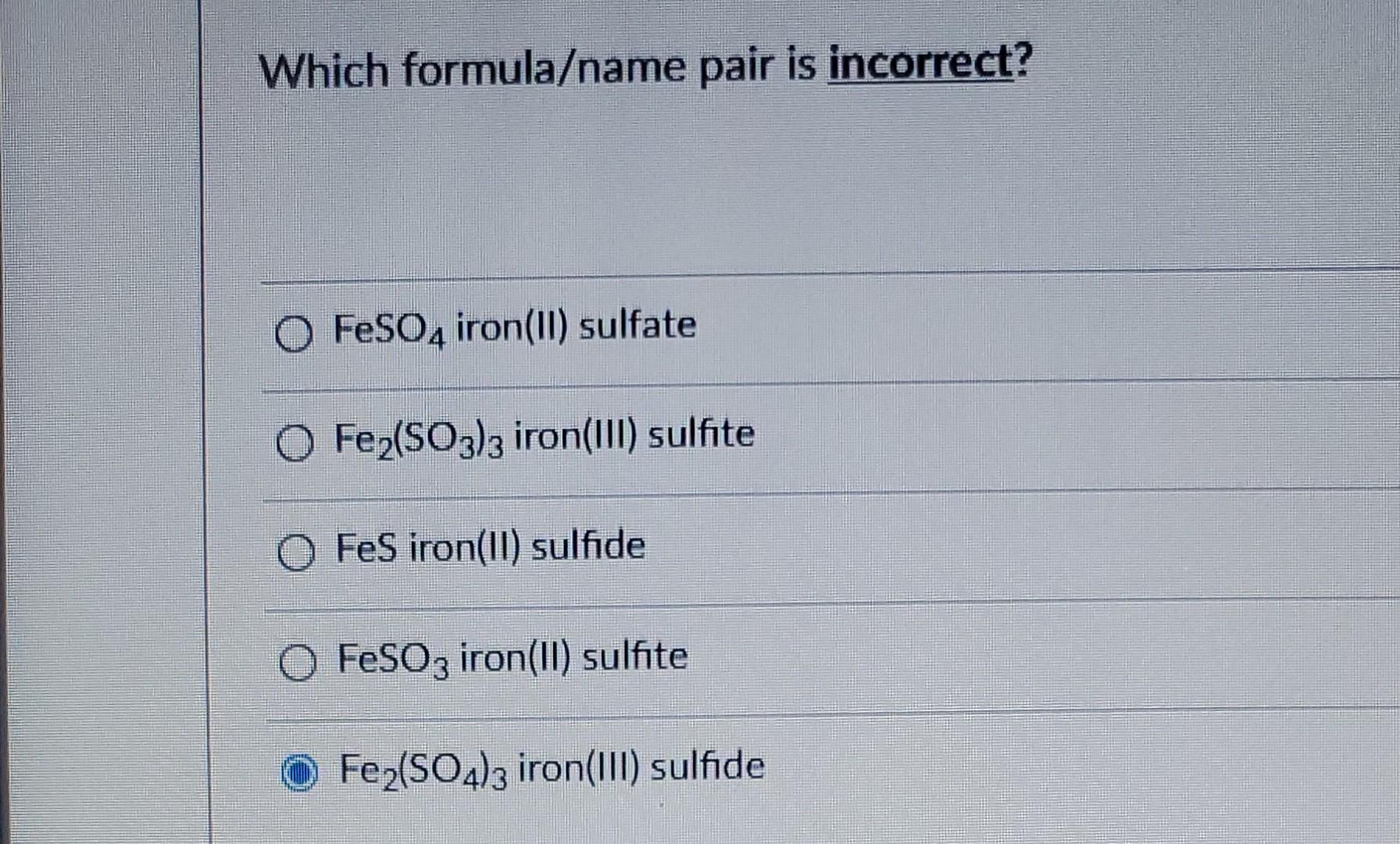
SOLVED: uestion 23 Which formula/name pair is incorrect? FeSO4 iron(Il) sulfate FeSO3 iron(Il) sulfite Fe2(SO3)3 iron(III) sulfite FeS iron(Il) sulfide Fez(SO4)3 iron(IlI) sulfide Moving to the next question prevents changes to this

SOLVED: Identify the type of reaction for each of the following as combination, decomposition, single replacement, double replacement, or combustion: (7.5) a. A compound breaks apart into its elements. b. Copper and