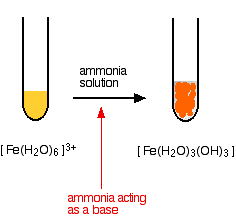
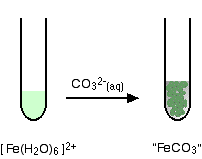
Fe(III) mobilisation by carbonate in low temperature environments: Study of the solubility of ferrihydrite in carbonate media and the formation of Fe( III) carbonate complexes - ScienceDirect

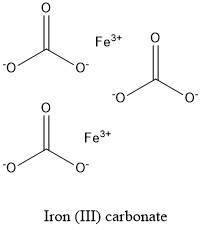
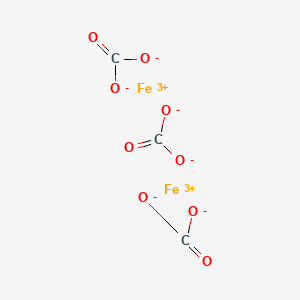
Oxidation Number for Fe2(CO3)3 . Oxidation state of fe2(co3)3 . Iron(III) carbonate oxidation state - YouTube
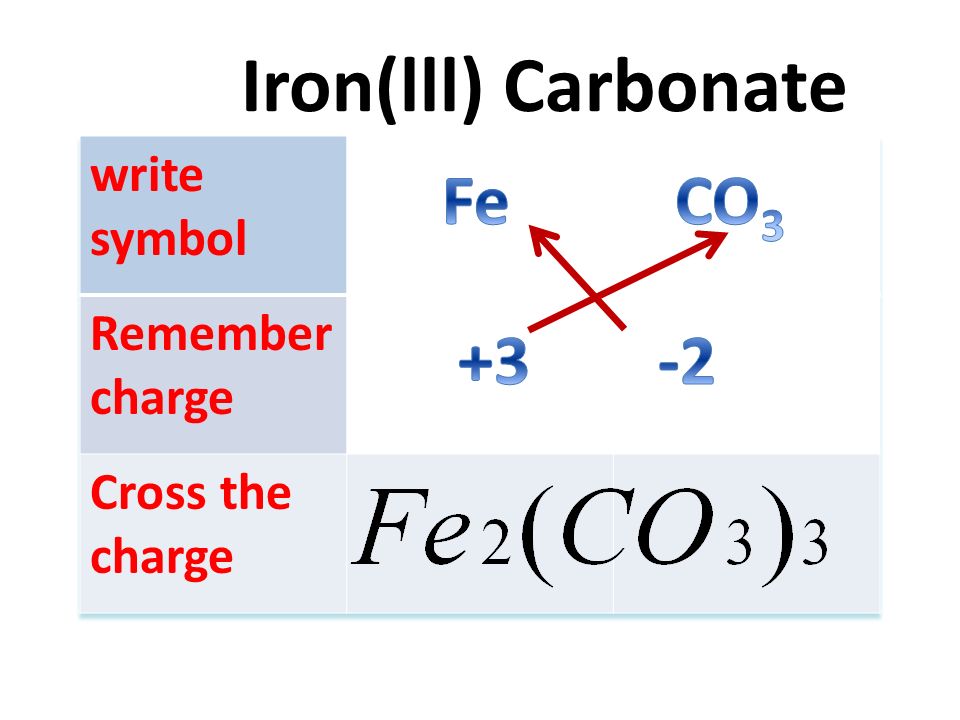
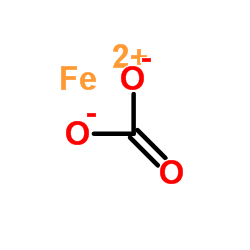
Negative ion Formula in compoundcharge Oxide Hydroxide Nitrate NO 3 -1 Sulphate Carbonate. - ppt download