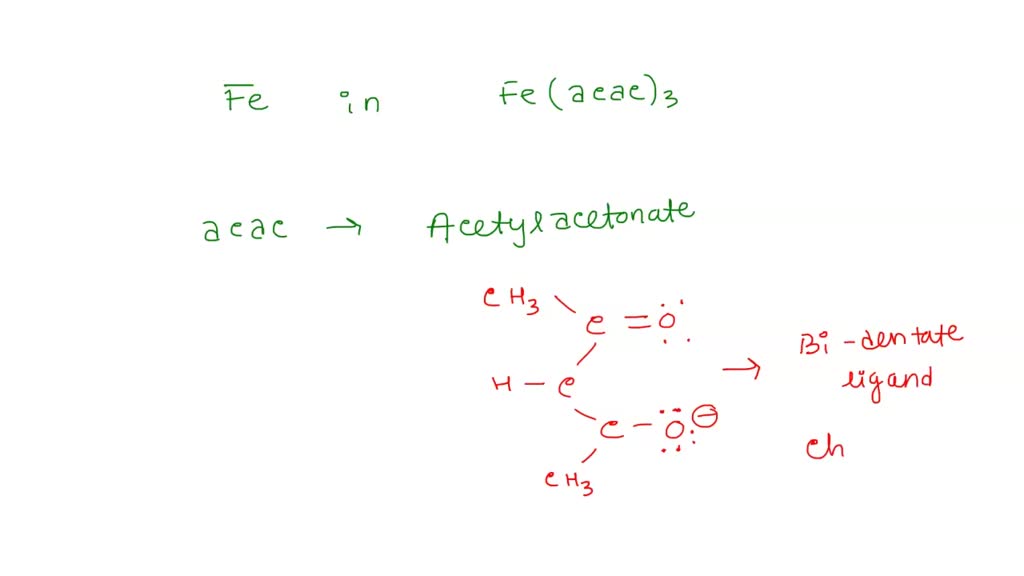![Prussian blue has two types of iron with oxidation number as shown below: IIIFe [ IIFe (CN)6 ] What is the net charge on prussian blue? Prussian blue has two types of iron with oxidation number as shown below: IIIFe [ IIFe (CN)6 ] What is the net charge on prussian blue?](https://d1hhj0t1vdqi7c.cloudfront.net/v1/YUpfOXpFZ19yYXM=/sd/)
Prussian blue has two types of iron with oxidation number as shown below: IIIFe [ IIFe (CN)6 ] What is the net charge on prussian blue?

Use of XPS in the determination of chemical environment and oxidation state of iron and sulfur samples: constitution of a data basis in binding energies for Fe and S reference compounds and

SOLVED: Determine the oxidation state for each of the elements below. iron(?) ion Fest The oxidation state of iron nitrous acid HNOz The oxidation state of nitrogen metaboric acid HBO2 The oxidation
The oxidation states of iron atoms in compounds (A), (B) and (C), respectively, are x, y and z. The sum of x,y and z is ..... - Sarthaks eConnect | Largest Online






![The oxidation state of iron in the brown ring complex [Fe(H2O)5NO]SO4 is : The oxidation state of iron in the brown ring complex [Fe(H2O)5NO]SO4 is :](https://d1hj4to4g9ba46.cloudfront.net/questions/1585570_1730433_ans_528a6827a2994b7486a15082e5ce4983.jpg)
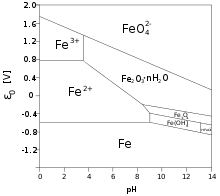
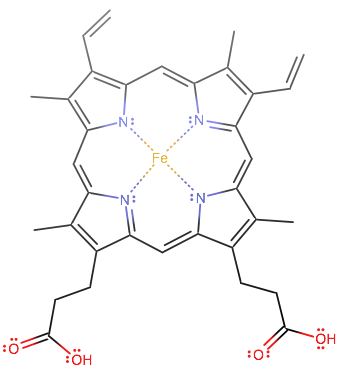





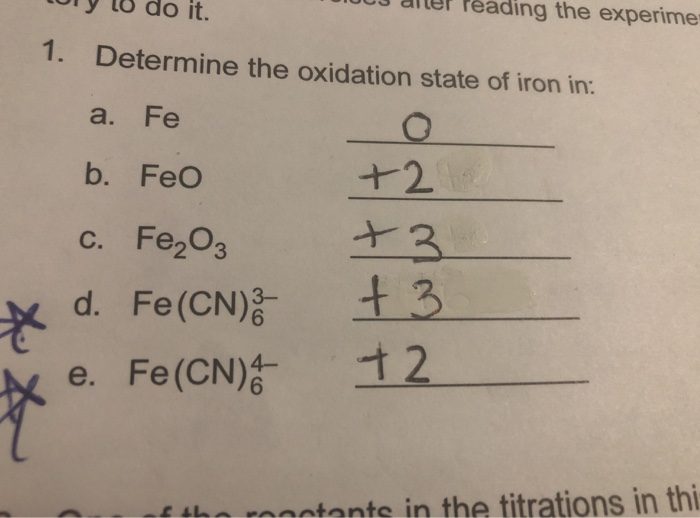
_Oxidation_States_for_First_Row_Transition_Metals.jpg?revision=1&size=bestfit&width=960&height=688)